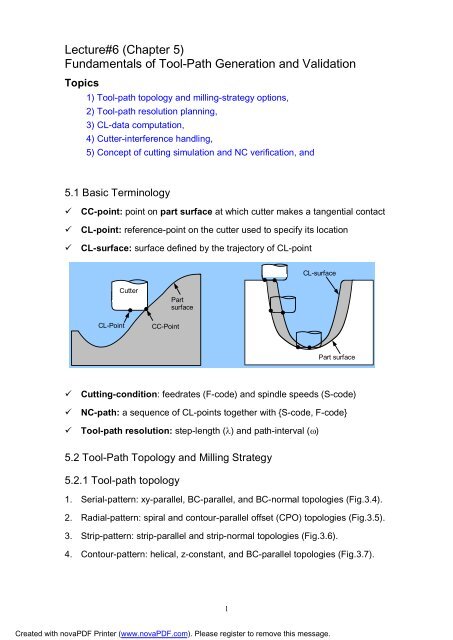
SOLVED: Use the data provided below to calculate the lattice energy of RbCl. Is this value greater or less than the lattice energy of NaCl? Explain. Electron affinity of Cl = -349

Novel Point of Care Rapid Coagulation Analyzer for Obstetrics – Quantra (SEER Sonorheometry) - ISTH Congress Abstracts

Given: E^oCr^3 + /Cr = - 0.74 V; E^oMnO4^ - / Mn^2 + = 1.51 V E^oCr2O7^2 + /Cr^3 + = 1.33 V; E^oCl/Cl^ - = 1.36 V Based on the data given above, the strongest oxidising agent will be: