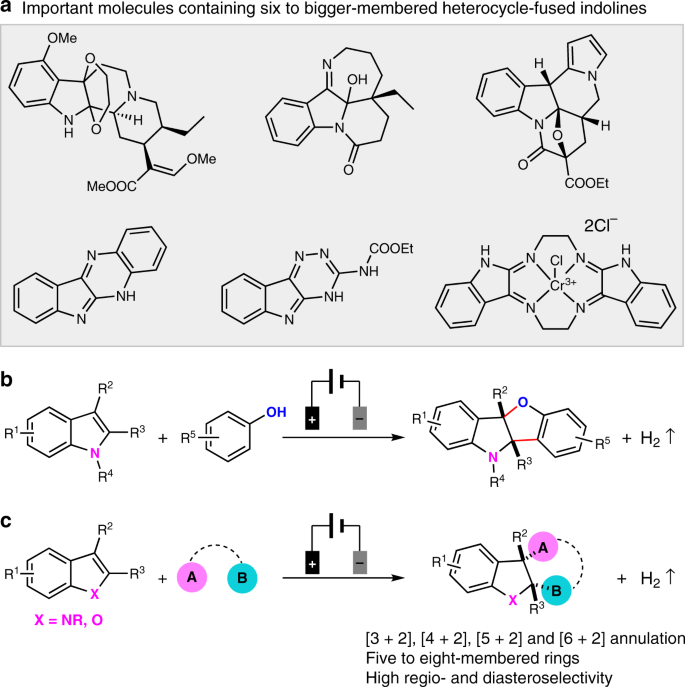
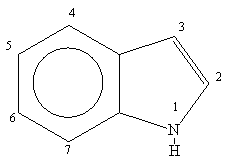
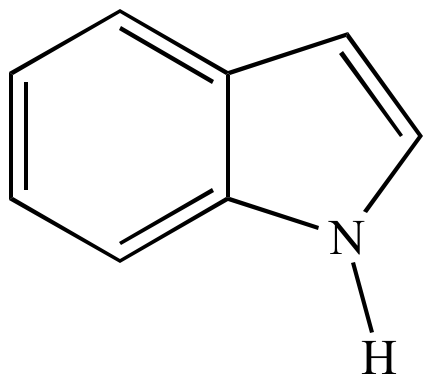
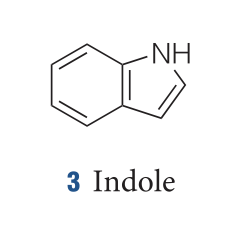
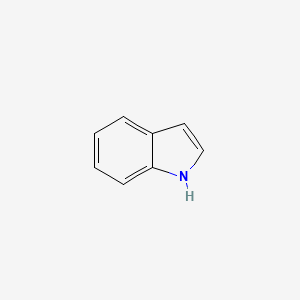
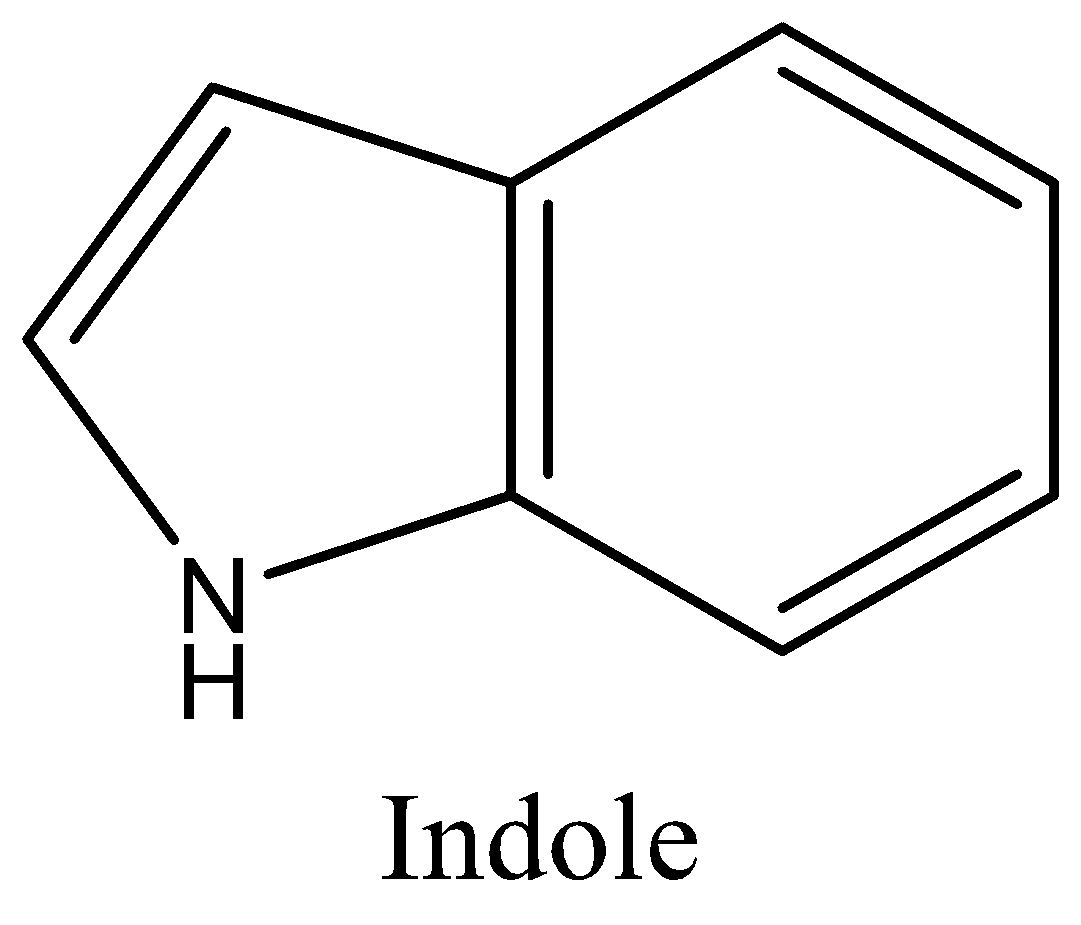
moleculestore on Twitter: "Happy #moleculemonday with @ministryofchemistry ・・・ #Indole is an #aromatic heterocyclic #organic compound with formula C8H7N. It has a bicyclic structure, consisting of a six-membered #benzene ring fused to a
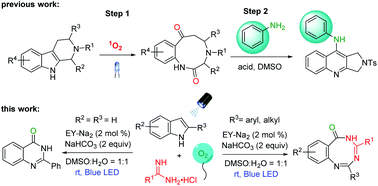
Visible-light-induced oxidative ring expansion of indoles with amidines - Organic Chemistry Frontiers (RSC Publishing)
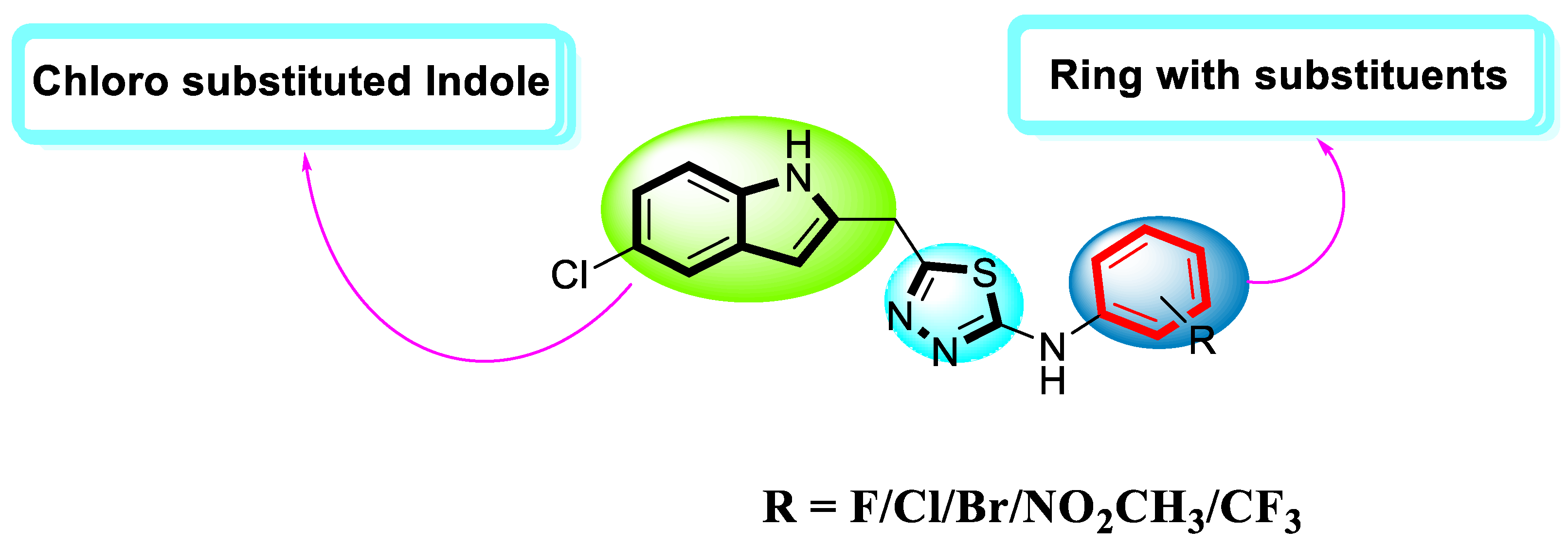
Molecules | Free Full-Text | Synthesis, In Vitro Biological Evaluation and In Silico Molecular Docking Studies of Indole Based Thiadiazole Derivatives as Dual Inhibitor of Acetylcholinesterase and Butyrylchloinesterase

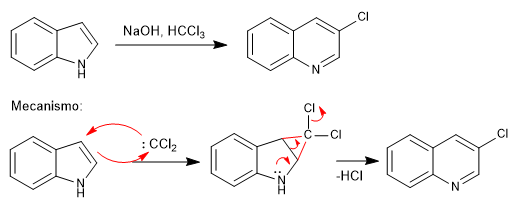
Carbon Atom Insertion into Pyrroles and Indoles Promoted by Chlorodiazirines | Journal of the American Chemical Society

Hypervalent Iodine Reagent for Introduction of Indoles via C-H activation of Arenes | TCI (Shanghai) Development Co., Ltd.

A) Structure of the perdeuterated indole ring-d 6 . The molecule is... | Download Scientific Diagram

Surface-enhanced Raman spectroscopy of indole ring-terminated self-assembled monolayer on silver electrode | Semantic Scholar

Metal-free ring expansion of indoles with nitroalkenes: a simple, modular approach to 3-substituted 2-quinolones - RSC Advances (RSC Publishing) DOI:10.1039/C4RA14406F

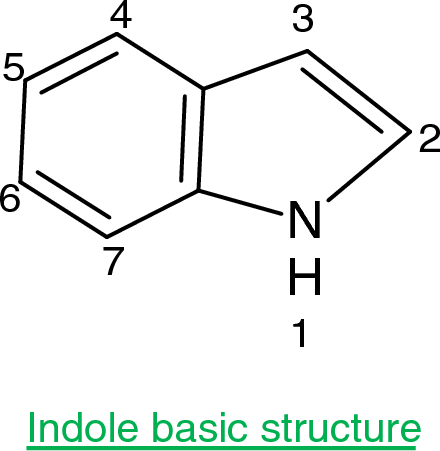
Indole is an aromatic heterocycle that has a benzene ring fused to a pyrrole ring. Draw an orbital picture of indole. (a) How many \pi electrons does indole have? (b) What is
The tryptophan molecule. The indole ring containing nitrogen, bottom... | Download Scientific Diagram

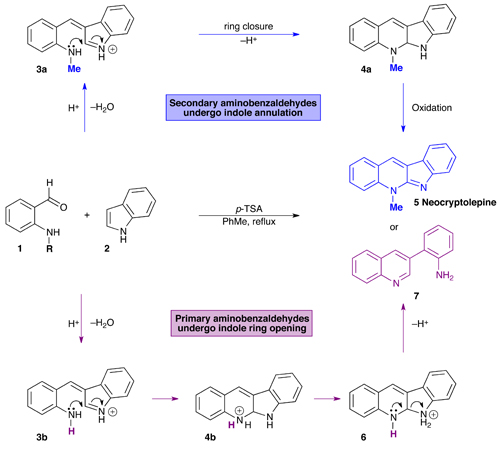
Synthetic Methods for 3,4‐Fused Tricyclic Indoles via Indole Ring Formation - Nemoto - 2018 - Asian Journal of Organic Chemistry - Wiley Online Library
Evaluation of the anti-inflammatory, analgesic, anti-pyretic and anti-ulcerogenic potentials of synthetic indole derivatives | Scientific Reports